Injectable vitamins have been voluntarily recalled by the manufacturer due to potential glass contamination, the U.S. Food and Drug Administration (FDA) has said.
One lot of Ascorbic Acid Solution for Injection produced by Nebraska-based STASKA Pharmaceuticals has been recalled “due to the presence of glass particulates in one lot of vials used in the production of this batch.”
The FDA release says that the product comes in 500mg/50mL vials and bears the lot code SP2400058, as well as an expiration date of December 31, 2024. There have been no adverse reactions or illnesses reported to date. It was distributed nationwide to licensed health-care providers between July 31 and August 27, 2024.
In a statement issued on the FDA website, STASKA Pharmaceuticals said it “places the utmost emphasis on patient safety and product quality.” The company added that it has “directly notified all customers who received the product by electronic mail and telephone and has arranged for the return and replacement of all recalled products.”
“Health care providers that have the product lot which is being recalled should stop using, quarantine, and contact STASKA Pharmaceuticals directly to arrange for return and replacement,” the company statement read.
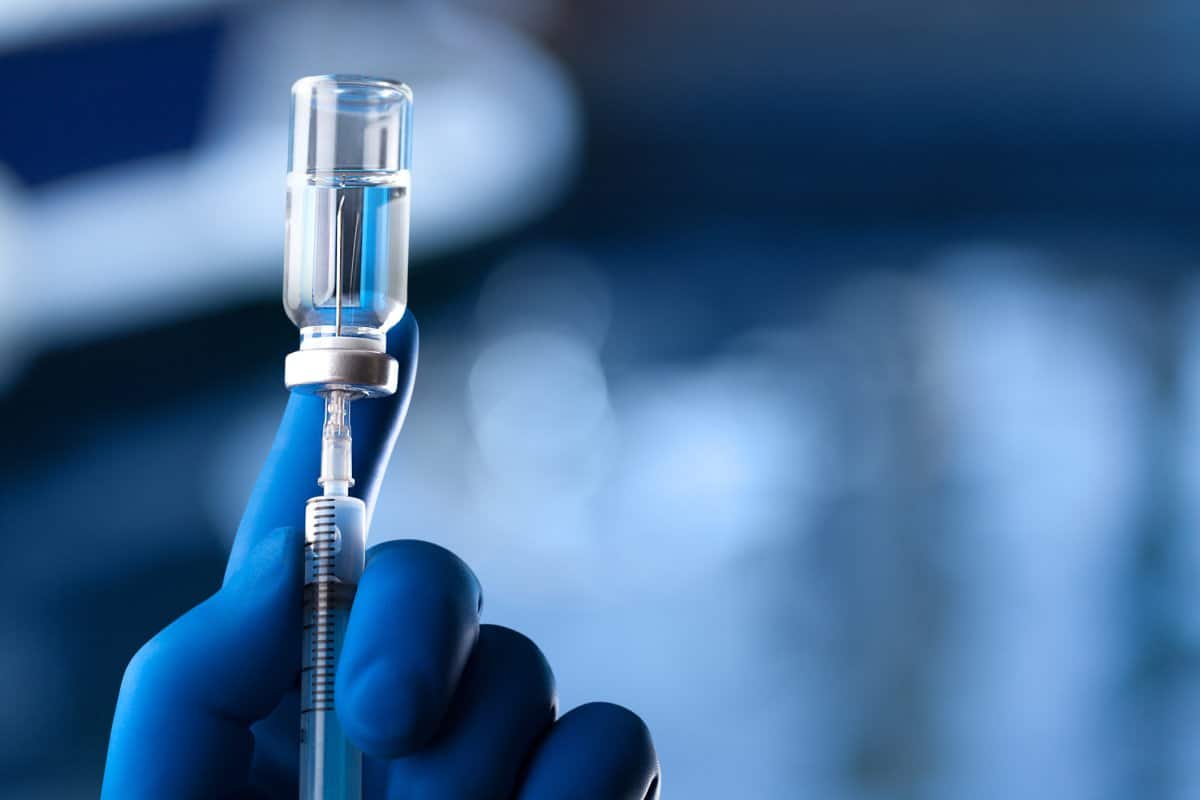
A hand in a glove holds an ampoule and a syringe. The injectable vitamins were found to contain glass particles.
GETTY
Administering an injectable product that contains particulate matter may lead to local irritation or swelling, the FDA said in the release. The particles can sometimes migrate through the body and obstruct blood vessels in critical areas such as the heart, lungs, or brain, resulting in serious complications like stroke and possibly death.
How serious these adverse effects can be is based on several factors, including the size and quantity of particles present in the medication; the patient’s existing health conditions; and whether any vascular anomalies are present.
Any consumers with questions regarding the recall should contact STASKA Pharmaceuticals on 402-782-2207 or 888-801-1370, Monday through Friday from 8 a.m. to 5 p.m. CT. Consumers should contact their physician or health-care provider if they have experienced any problems that may be related to taking or using this drug product.
In September, batches of antiviral drug Veklury, which is also administered intravenously, were recalled for the same reason after a customer complaint. The drug is used to treat adults and children who have contracted COVID-19 and are seriously ill or at risk of becoming seriously ill. Veklury, also known as remdesivir, is not available in stores and can be administered by only health-care professionals.
Another recall was issued in March this year for the drug Methocarbamol, which is produced by Eugia US LLC, as it contained “white particles” floating inside of the vial. Methocaramol is used to treat treats muscle pain, spasms and stiffness.









